Article Summary by Robert Skinner, MD
Background:
The leading cause of death in the United States for individuals under the age of 40 is trauma, according to the CDC, with an estimated death toll of greater than 160,000 people per year [1]. Because of this, attention has been placed on how to improve trauma outcomes in the prehospital setting. One area of interest in this field is on the deployment of EMS physicians, and the practicality of their presence in the prehospital field.
The practice of using prehospital EMS physicians is somewhat routine in many placed in Europe [2], however is still somewhat novel in the United States. This study points out that little literature had shown a benefit of the presence of EMS physicians in the prehospital setting, despite this being a listed key priority in Emergency and Prehospital Medicine research. This study performed a systematic literature review and meta-analysis to look at the mortality levels in severely injured patients treated by physicians in the prehospital setting compared to those treated by a paramedic led team.
Methods:
A review of the literature was performed using PubMed and Google scholar, reviewing articles published up to 2018. The authors mention that a hand search of their personal literature was performed as well. Search criteria included studies reporting mortality or survival of severely injured patients treated either by EMS physicians or by the traditional paramedic led team. Inclusion criteria included any study discussing patients suffering from an acute traumatic injury. Only those studies with a comparative element were included in this study. This includes randomized control trials, matched-pairs analysis, before and after design or observation studies with a comparative element. Scores mentioned used to compare characteristics include the Injury Severity Score (ISS), Abbreviated Injury Scale (AIS), and predicted mortality. Included studies were compared using fixed effect and random effects meta-analysis using inverse variance weighting for pooling. To account for any potential changes in care regarding ground transport vs air ambulance, a subgroup analysis was performed on studies where the effect of helicopter transport could be excluded (i.e studies with no helicopter transport in either the physician group or paramedic group or studies that had helicopter transport in both groups.)
Results:
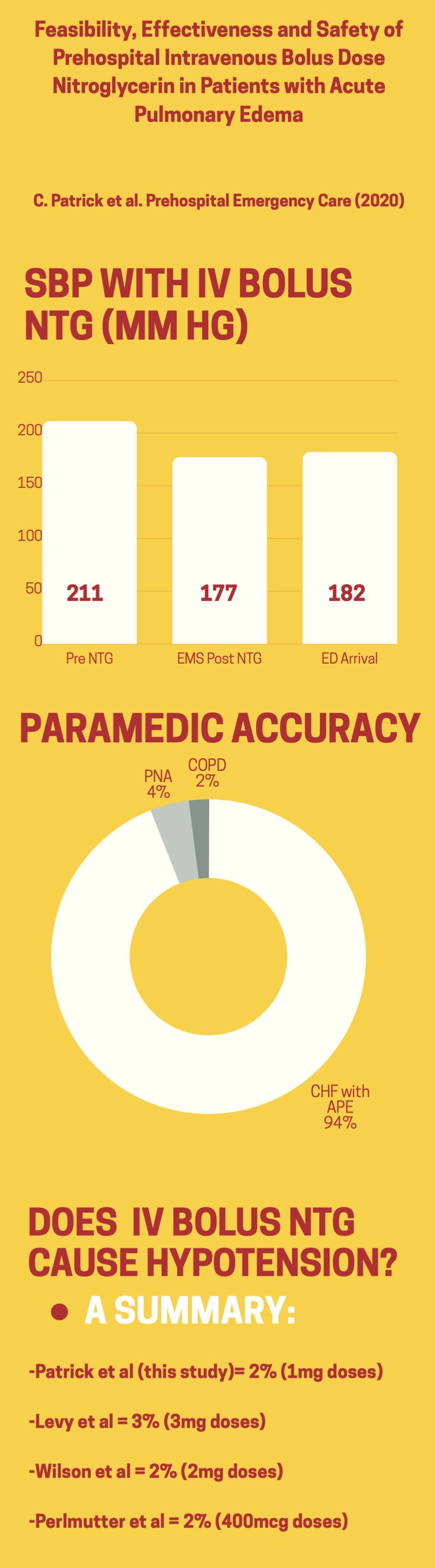
A total of 2,249 publications were considered for inclusion in this study, and after exclusion criteria was applied, a total of 22 studies were considered eligible for the analysis. The included studies had a pooled sample size of 54,991 patients suffering from what was considered a severe injury. 13,629 patients were treating by a team including an EMS physician, and 41,362 were treated by a traditional paramedic led team without an on scene physician. The study reports that in the overall analysis of all included studies, the odds ratios for mortality were significantly lower in the EMS physician group (OR of 0.81, 95% CI 0.71-0.92) compared to a team without EMS physician presence.
When the analysis was subdivided to only include studies that were adjusted or matched for injury severity using one of previously mentioned scores (in other words, where injury scores between the two studies would be comparable), the OR was 0.86 (95% CI 0.73-1.01), making the result not statistically significant. When only more recent studies (2005-2018) were included, the OR was 0.75 (95% CI 0.64-0.88) for all studies and 0.81 (95% CI 0.67-0.97) in studies adjusted for injury severity.
In the subgroup analysis of studies with comparable modes of transport (to eliminate confounder of helicopter transport), when comparing EMS physician led treatment to paramedic based, the OR for mortality was 0.80 (95% CI 0.65-1.00) in all studies and 0.74 (95% CI, 0.53–1.03) in the more recent studies. Although not statistically significant, there is a trend towards clinical significance.
What does this study mean for EMS?
This study suggests that there may in fact be a survival benefit for severely injured trauma patients when an EMS physician is on scene and part of the treatment team. The sample size is impressive, with 22 international studies included and a patient population of greater than 54,000 patients. Although the date of 2005 may seem somewhat arbitrary as a cutoff for “older vs newer” literature, the study does note that training in prehospital trauma management has significantly improved over the past decade, and comparing newer studies to evaluate for changes in standards of care certainly seems to strengthen this study.
Some limitations in the study include the fact that the majority of the studies included were retrospective and observational, and the fact that timing of the mortality benefit varied by study (as most studies measured mortality after 30 days or in hospital, however some measured mortality up to a year.)
EMS clinicians deliver excellent care patient care. When available; EMS physician presence seems to be beneficial as part of a team led effort for those patients who are severely injured. Additional work and research should be pursued in this topic.
References
1. Centers for Disease Control and Prevention. National Center for Health Statistics. Leding Causes of Death. https://www.cdc.gov/nchs/fastats/ leading-causes-of-death.htm. Updated March 17, 2017. Accessed March 22, 2019.
2. Wilson MH, Habig K, Wright C, Hughes A, Davies G, Imray CH. Prehospital emergency medicine. Lancet. 2015;386(10012):2526–2534.
3. Fevang E, Lockey D, Thompson J, Lossius HM, Torpo Research Collabora- tion. The top five research priorities in physician-provided pre-hospital critical care: a consensus report from a European research collaboration. Scand J Trauma Resusc Emerg Med. 2011;19:57.
Website Editing and Layout by EMS MEd Editor James Li MD